Is life-changing NeuroStar® TMS Therapy a good fit for you? Take our quiz to find out!

When other depression treatments have failed, there is hope!
New Directions Mental Health is proud to partner with NeuroStar® to provide Transcranial Magnetic Stimulation (TMS) as an option for our clients. TMS is a safe, effective, non-drug treatment for major depression without the common side effects of medication. It is FDA-cleared to treat depression, depression with anxiety, and OCD in adults, and it is now FDA-cleared to treat adolescents aged 15-21 as an add-on therapy. Most major insurance plans cover NeuroStar® TMS, including Medicare plans nationwide.
Mark George M.D. Biological Psychiatry Branch Division of Intramural Research Progress, NIMH 1993
NeuroStar® TMS Therapy FAQ
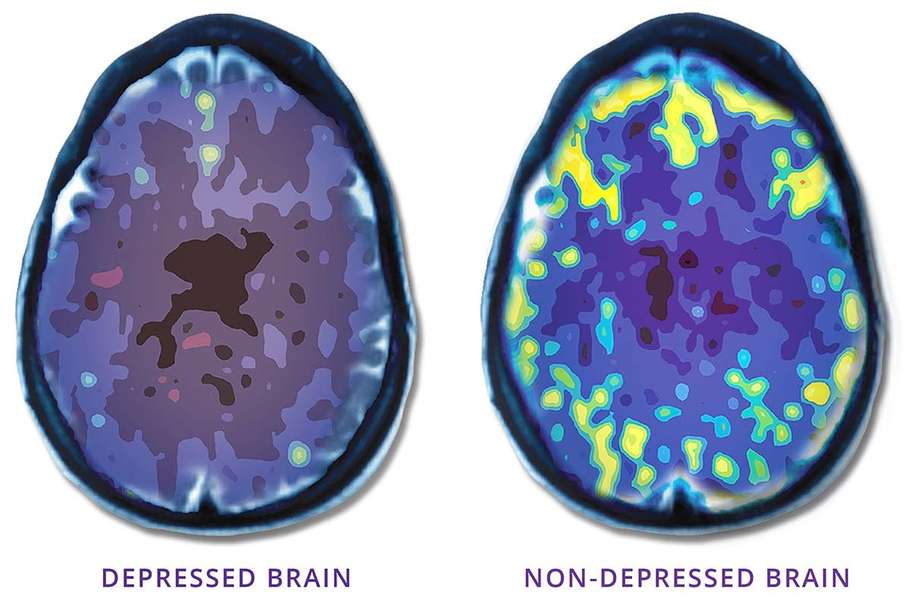
NeuroStar® TMS uses focused magnetic pulses (similar in strength to an MRI), to revitalize underactive areas of the brain involved in regulating mood. When these regions are underactive, depression can result. “Waking up” these regions can have a lasting effect on depression, making long-term remission from depression a reality for many people.
A real-world study reported an 83% response rate. This means that 83% of patients that completed their NeuroStar® TMS treatment cycle saw measurable improvement in their depression symptoms. The same real-world study reported a 62% remission rate. This means that 62% of patients that completed their NeuroStar® TMS treatment cycle likely wouldn’t be diagnosed with MDD if they were first being evaluated by their doctor.
Yes, NeuroStar® TMS is a safe and FDA-cleared treatment for depression, depression with anxiety, and OCD. More than 6.4 million NeuroStar® TMS therapy treatments have been performed in over 175,000 patients, and less than five percent of clients reported any side effects. The most common side effect is a headache right after NeuroStar® TMS treatment, which goes away after the first week of sessions for most people.
Many patients experience side effects from their medication, increasing the chance that they will stop taking them. The most common side effect with NeuroStar® is mild to moderate pain or discomfort at the treatment site. For most patients this subsides within the first week of treatment.
Most major insurances cover NeuroStar® TMS, including Medicare plans nationwide.
Adults aged 22-70. Adolescents aged 15-21 are eligible for TMS as an add-on therapy.
NeuroStar® TMS is FDA-cleared for the treatment of Major Depressive Disorder (MDD) in adults 22-70, as an add-on therapy for adolescents aged 15-21, and for the treatment of Obsessive Compulsive Disorder (OCD) in adults aged 22-70.
NeuroStar TMS treatment is conducted in one of our New Directions Mental Health clinics. You will meet with our team for a consultation to determine your eligibility. Depending on your provider’s recommendation, a typical TMS treatment plan includes 36 sessions that each last between 19-37 minutes. Completing the full treatment course gives the best chance for delivering long-lasting relief from depression.
- Before Treatment: You’ll recline comfortably in the treatment chair. A curved magnetic coil will be positioned on your head.
- During Treatment: You’ll hear a clicking sound and feel a tapping sensation on your head as the NeuroStar® coil focuses treatment precisely at the source of the depression: the brain. The patented Contact Sensing technology ensures the prescribed dose is delivered, every treatment, every pulse.
- After Treatment: With NeuroStar® TMS you are fully awake and alert during treatment and may drive home and resume daily activities immediately afterwards. There are no negative effects on memory or sleep.
What to Expect
TMS treatment is conducted in one of our New Directions Mental Health clinics. You will meet with our team for a consultation to determine your eligibility. Depending on your provider’s recommendation, a typical TMS treatment plan includes 36 sessions. Completing the full treatment course gives the best chance for delivering long-lasting relief from depression.

Step 1: Before Treatment
You’ll recline comfortably in the treatment chair. A small, curved magnetic coil will be positioned lightly on your head. Our dedicated team members will help you through this process.

Step 2: During Treatment
NeuroStar® TMS delivers focused magnetic stimulation directly to the target areas of the brain. You’ll hear a clicking sound and feel a tapping sensation on your head, but don’t worry, for most patients, any pain or discomfort is only temporary and typically resolves within the first week of treatment.

Step 3: Begin your journey
Depending on your doctor’s recommendation, you can resume normal activities immediately. Because there are no effects on alertness, you can drive yourself to and from treatment sessions. In-office treatment with NeuroStar® TMS Therapy typically takes 19-37 minutes and treatment takes place every day during the week for 7 weeks.
Discover how NeuroStar® TMS treatment can help you or a loved one today.
The Support You Need, When You Need It
About New Directions Mental Health
New Directions Mental Health is dedicated to transforming lives by offering quality, easily accessible mental health care in Southwest Pennsylvania. We believe access to quality mental health care is a right, not a privilege and are dedicated to connecting clients to care in a way that works for their individual needs. That is why we accept most major insurance plans, including Medicaid and Medicare, and we are deeply rooted in our communities. We offer in-person services at our local clinics, as well as convenient telehealth options.
TMS Testimonials




The NeuroStar® Advanced Therapy System is indicated for the treatment of depressive episodes and for decreasing anxiety symptoms for those who may exhibit comorbid anxiety symptoms in adult patients suffering from Major Depressive Disorder (MDD) and who failed to achieve satisfactory improvement from previous antidepressant medication treatment in the current episode. The NeuroStar® Advanced Therapy system is intended to be used as an adjunct for the treatment of adult patients suffering from Obsessive-Compulsive Disorder (OCD). NeuroStar® Advanced Therapy is only available by prescription. A doctor can help decide if NeuroStar® Advanced Therapy is right for you. Patients’ results may vary. Visit neurostar.com for full safety and prescribing information.
Adult Indications for Use
The NeuroStar® Advanced Therapy System is indicated for the treatment of depressive episodes and for decreasing anxiety symptoms for those who may exhibit comorbid anxiety symptoms in adult patients suffering from Major Depressive Disorder (MDD) and who failed to achieve satisfactory improvement from previous antidepressant medication treatment in the current episode.
The NeuroStar® Advanced Therapy System is intended to be used as an adjunct for the treatment of adult patients suffering from Obsessive-Compulsive Disorder (OCD).
Adolescent Indications for Use
NeuroStar® Advanced Therapy is indicated as an adjunct for the treatment of Major Depressive Disorder (MDD) in adolescent patients (15-21).
NeuroStar® Advanced Therapy is only available by prescription. A doctor can help decide if NeuroStar® Advanced Therapy is right for you. Patients’ results may vary.
Important Safety Information
The most common side effect is pain or discomfort at or near the treatment site. These events are transient; they occur during the TMS treatment course and do not occur for most patients after the first week of treatment. There is a rare risk of seizure associated with the use of TMS therapy (<0.1% per patient).
NeuroStar® Advanced Therapy should not be used with patients who have non-removable conductive metal in or near the head. NeuroStar® Advanced Therapy has not been studied in patients who have not received prior antidepressant treatment.
Your Journey to Mental Well-Being Starts Here
Take the courageous first step toward a new chapter in your life. Reach out for mental health treatment in your area today by completing our brief questionnaire to connect with the compassionate professionals at New Directions Mental Health.
Our Locations
South Hills, PA
180 Fort Couch Road,
Suite 304
Pittsburgh, PA 15241
Wexford, PA – VIP Drive
117 VIP Drive,
Suite 310
Wexford, PA 15090
Monongahela, PA
1051 Country Club Rd
Monongahela, PA 15063
South Side Flats, PA
Coming Soon!
2516 Jane Street
Suites 200 and 205
Pittsburgh, PA 15203
Robinson, PA
3 Robinson Plaza,
#340
Robinson, PA 15205
Washington, PA
640 Jefferson Ave
Washington, PA 15301
Greensburg, PA
420 Pellis Rd
Greensburg, PA 15601
Meadowlands, PA
3000 Park Place Drive,
Suite 108
Washington, PA 15301
Monroeville, PA
4055 Monroeville Blvd
Monroeville, PA 15146
Fox Chapel, PA
1000 Gamma Dr.
Suite 501
Pittsburgh, PA 15238
Wexford, PA – Stonewood
7000 Stonewood Drive,
Suite 300
Wexford, PA 15090
McMurray, PA
110 Hidden Valley Road
McMurray, PA 15317
© Copyright 2023 Transformations Care Network | 724-237-4691










